EULAR Research Consortia Partnerships
EULAR welcomes consortia committed to advancing rheumatology research through meaningful patient involvement, strategic collaboration, and alignment with European research priorities. We partner with research consortia that demonstrate strong scientific leadership and actively include patients from the earliest stages of international research initiatives.
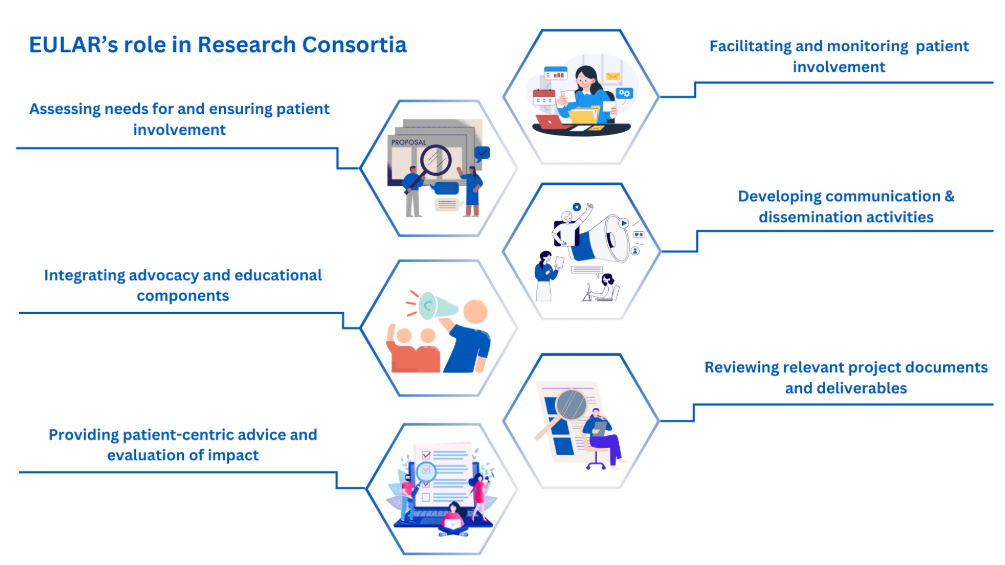
EULAR collaborates with EU-funded research consortia to:
- Support excellence and competitiveness in rheumatology research.
- Promote transnational, interdisciplinary collaboration by serving as a central hub and fostering synergies across European research teams and projects.
- Provide expert guidance in designing, planning and implementing collaborative research, including effective patient-researcher partnerships.
- Ensure structured, inclusive and meaningful patient involvement through access to the EULAR network of trained Patient Research Partners (PRPs).
- Offer information on EULAR’s infrastructure and resources, including funding schemes, advocacy, scientific events, education and training opportunities.
- Support outreach, communication and dissemination efforts by enabling consortia to connect with EULAR’s broad network of health professionals and patients.
Eligibility
EULAR is interested in partnering with research consortia that meet the following criteria:
- Rheumatology leadership: The consortium and its research topic should be rheumatology-led or co-led.
- Consortium leadership: The consortium should be led by academic partners or other non‑commercial institutions.
- Geographic scope: Most consortium partners should be based in EULAR-affiliated countries.
- External funding: The consortium should apply for funding from sources external to EULAR (e.g., Horizon Europe, Innovative Health Initiative).
- Full partnership: EULAR should be formally recognised as a full partner in the consortium. Depending on the agreed role, EULAR will determine the budget required to deliver its activities.
- Early engagement: The consortium should contact EULAR as early as possible during proposal development and no later than eight weeks before the submission deadline.
- Patient involvement from the outset: The consortium should ensure the active and equitable involvement of PRPs from the earliest stages of project design.
- Strengthening local involvement: The consortium should support the recruitment of local PRPs for the project and contribute to strengthening local PRP networks.
- Adherence to EULAR standards: The consortium should commit to following the EULAR recommendations for involving PRPs in rheumatology research, as well as the associated reference cards for collaborative research.
Request a consortia collaboration here
Please note that we are no longer accepting partnership requests for proposals for Horizon Europe calls due on 16 April or for Innovative Health Initiative calls due on 21 April.
Terms and Conditions
Support from EULAR is subject to organisational capacity and the degree to which the proposed project aligns with EULAR’s mission and community. Due to a high volume of enquiries, response times may vary; early contact with the EULAR Research Team is strongly recommended to maximise the likelihood of collaboration.
EULAR reserves the right to withdraw from a partnership if the consortium fails to comply with the established eligibility requirements.
Research Ethics
EULAR expects the highest standards in all the research it supports. This includes strict scientific integrity and full adherence to recognised principles of good research practice. Any involvement of patients in research must comply with the Declaration of Helsinki and ensure that all participants are treated with dignity, safety, and respect.
Ongoing Consortia Partnerships

PerPrev-CID
Time frame: 2025 - 2029
Advancing Personalised Prevention and Early Intervention for Chronic Inflammatory Diseases


AutoPiX
Time frame: 2024 - 2029
Imaging for Patient Benefit in Arthritis
AUTOPIX

STRATA-FIT
Time frame: 2023 - 2029
Stratification of Rheumatoid Arthritis: Computational models to personalize management strategies for difficult-to-treat disease

SQUEEZE
Time frame: 2022 - 2027
Maximizing Impact of Prescription Drugs in Rheumatoid Arthritis

HIPPOCRATES
Time frame: 2021 - 2026
Promoting Early Identification and Improving Outcomes in Psoriatic Arthritis
Previous Consortia Partnerships

HarmonicSS
Time frame: 2017 - 2020
HARMONIzation and integrative analysis of regional, national and international cohorts on primary Sjögren's Syndrome (pSS) towards improved stratification, treatment and health policymaking
EULAR support for Research Consortia
Related Information
Sign Up to the EULAR Research Newsletter
To keep up to date with EULAR and developments around the EULAR Research Centre, sign up to receive the Newsletter here.
Questions?
Contact the Research team at research@eular.org